|
The simplest of these structures are micelles, spherical assemblies containing a hydrophobic interior of phospholipid tails and an outer surface of polar head groups. As a result, energetically stable, large-scale assemblies of phospholipid molecules are formed in which the hydrophobic tails congregate within enclosed regions, shielded from contact with water by the polar heads (Figure 3). Because of their considerable lengths, these tails are, in fact, strongly attracted to one another. As mentioned, the polar heads of these molecules are strongly attracted to water molecules, and the nonpolar tails are not. The amphipathic nature of phospholipids enables them to form uniquely functional structures in aqueous environments. Glycolipids are examples in which carbohydrates are bonded to the lipids’ head groups. Notice the “R” designation within the hydrophilic head depicted in Figure 2, indicating that a polar head group can be more complex than a simple phosphate moiety. Because the phosphate is charged, it is capable of strong attraction to water molecules and thus is hydrophilic, or “water loving.” The hydrophilic portion of the phospholipid is often referred to as a polar “head,” and the long hydrocarbon chains as nonpolar “tails.” A molecule presenting a hydrophobic portion and a hydrophilic moiety is said to be amphipathic. Conversely, phospholipids such as the one shown in Figure 2 have a negatively charged phosphate group. Because all three substituents on the glycerol backbone are long hydrocarbon chains, these compounds are nonpolar and not significantly attracted to polar water molecules-they are hydrophobic. Figure 1 depicts the structure of a triglyceride. The molecular structure of lipids results in unique behavior in aqueous environments. Instead of another fatty acid molecule (as for triglycerides), the third binding position on the glycerol molecule is occupied by a modified phosphate group. The two fatty acid carbon chains may be both saturated, both unsaturated, or one of each. Figure 2 depicts a typical phospholipid composed of two fatty acids linked to glycerol (a diglyceride). In contrast, complex lipids contain at least one additional component, for example, a phosphate group ( phospholipids) or a carbohydrate moiety ( glycolipids). Triglycerides are classified as simple lipids because they are formed from just two types of compounds: glycerol and fatty acids. The unsaturated fatty acid has a slight kink in its structure due to the double bond. This illustration shows a phospholipid with two different fatty acids, one saturated and one unsaturated, bonded to the glycerol molecule. They play an important metabolic role, serving as efficient energy-storage molecules that can provide more than double the caloric content of both carbohydrates and proteins.įigure 2. Triglycerides are the primary components of adipose tissue (body fat), and are major constituents of sebum (skin oils). For example, lipids containing saturated fatty acids are solids at room temperature, whereas lipids containing unsaturated fatty acids are liquids.Ī triacylglycerol, or triglyceride, is formed when three fatty acids are chemically linked to a glycerol molecule (Figure 1). These differences in saturated versus unsaturated fatty acid structure result in different properties for the corresponding lipids in which the fatty acids are incorporated. Saturated fatty acids have a straight, flexible carbon backbone, whereas unsaturated fatty acids have “kinks” in their carbon skeleton because each double bond causes a rigid bend of the carbon skeleton. Fatty acids with hydrocarbon chains containing at least one double bond are called unsaturated fatty acids because they have fewer hydrogen atoms. Fatty acids with hydrocarbon chains that contain only single bonds are called saturated fatty acids because they have the greatest number of hydrogen atoms possible and are, therefore, “saturated” with hydrogen. Because the long hydrocarbon chain, fatty acids are hydrophobic (“water fearing”) or nonpolar. The fatty acids are lipids that contain long-chain hydrocarbons terminated with a carboxylic acid functional group. Lipids comprise a broad class of many chemically distinct compounds, the most common of which are discussed in this section. They can be a source of nutrients, a storage form for carbon, energy-storage molecules, or structural components of membranes and hormones. Lipids serve numerous and diverse purposes in the structure and functions of organisms. Describe how phospholipids are used to construct biological membranes.Īlthough they are composed primarily of carbon and hydrogen, lipid molecules may also contain oxygen, nitrogen, sulfur, and phosphorous.Compare and contrast triacylglycerides (triglycerides) and phospholipids.Describe the unique characteristics and diverse structures of lipids. 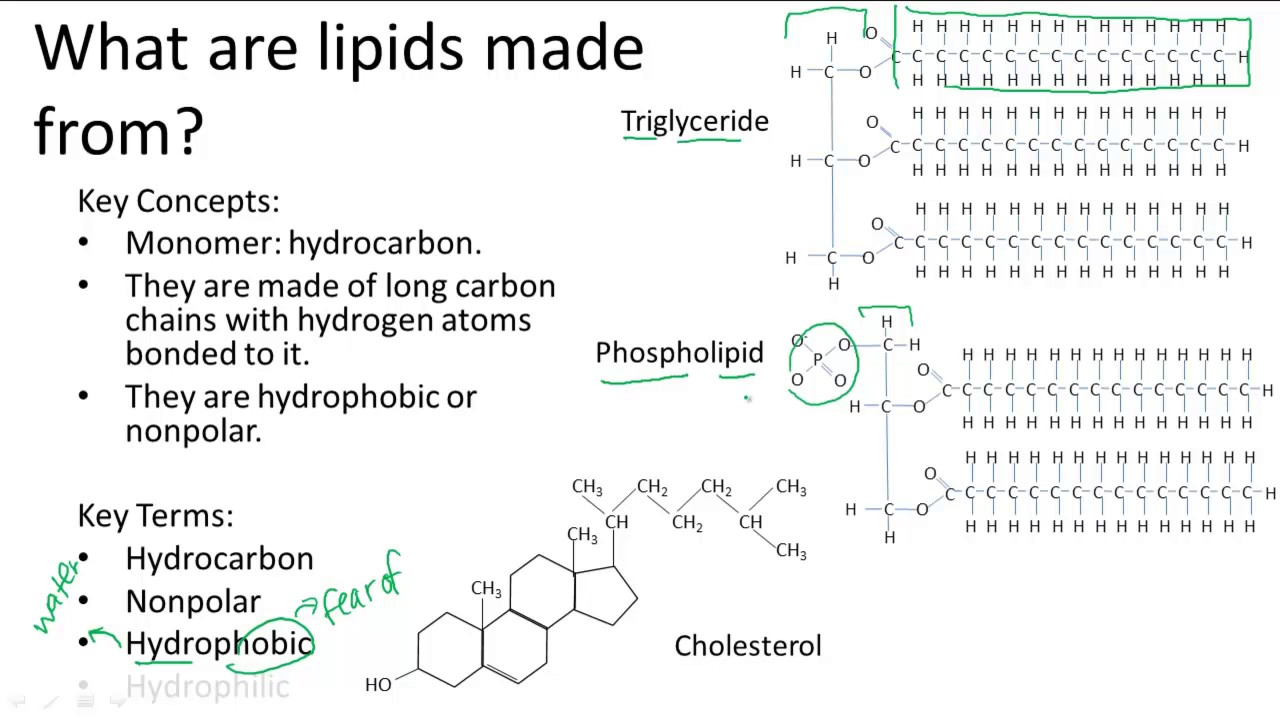
Describe the chemical composition of lipids
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
